Research
When Mary Free Bed Rehabilitation Hospital conducts research, it’s not only for the future benefit of science and medicine. Our team is dedicated to generating evidence-based, innovative treatment and applying it as quickly as possible to help people recovering from serious injuries or illnesses.
The research efforts of our doctors, therapists, nurses and other professionals often lead to discoveries with a timely and positive impact for our patients. Research is done with the conviction it will help restore hope and freedom for children and adults with physical disabilities.
Mary Free Bed has created an infrastructure through the John F. Butzer Center for Research and Innovation to support our physicians and staff with research endeavors and to facilitate scholarly activity. Our efforts include original research, participation in multicenter studies and sponsored clinical trials.
Study inquiries or requests
Email: research@maryfreebed.com
Call: 616.840.8926
Current Studies
Active recruitment opportunities include:
Sleep for Stroke Management and Recovery Trial (Sleep SMART)
Mary Free Bed is participating in the Stroke Net study Sleep SMART. This 110-site study randomizes participates who have sleep apnea to test whether treatment of obstructive sleep apnea with continuous positive airway pressure (CPAP) is effective for secondary prevention and recovery after stroke. Eligible participants must be referred from acute care and receiving inpatient therapeutic services for a stroke diagnosis. For more information, click here.
Principal Investigator: Devin L. Brown, M.D., MS (University of Michigan)
Bone Health Biobank
Researchers are collecting saliva samples from Mary Free Bed patients who have a bone health diagnosis. Genetic mapping will be done to assess relationships with specific gene profiles and diagnoses. Results may contribute to changes in clinical practice and treatment of diagnoses, such as osteoarthritis and osteopenia. Eligible participants must have a bone-related diagnosis and be treated at Mary Free Bed.
Principal Investigator: Teri Holwerda, NP
Defining Trajectories of Linguistic, Cognitive-Communicative and Quality of Life Outcomes in Aphasia
The objective of this study is to describe the trajectories of linguistic, cognitive-communicative and health-related quality of life outcomes following stroke in people with aphasia during inpatient and outpatient rehabilitation to 18 months following their stroke. A sample of 300 consecutively admitted stroke patients with aphasia recruited at three Midwest rehabilitation hospitals will complete measures of linguistic and cognitive-communicative performance and the Quality of Life Neurological Disorders Measurement instruments during rehabilitation and at three-, six-, 12- and 18-months post stroke. We will model outcomes as individual and group trajectories, allowing us to develop individual predictions that could inform clinical planning and decision-making for new patients. For more information, click here.
Principal Investigators: Leora Cherney, Ph.D. (Shirley Ryan AbilityLab); Allen Heinemann, Ph.D. (Shirley Ryan AbilityLab)
Post-marketing, Randomized Crossover Study to Evaluate the Clinical Safety and Efficacy of Upper Limb Spasticity Treatment
Researchers are seeking robust evidence to enable informed therapeutic decisions for care and cost optimization, including use of optimal dose.
Principal Investigator: Dr. Jonathan Vandenberg, M.D.
MI-CHAP
Mary Free Bed Rehabilitation Hospital, in partnership with Michigan State University’s College of Human Medicine, has been awarded funding from the Michigan Health Endowment Fund under its Healthy Aging initiative.
The fund supports projects that improve access to care, allow Michigan residents to age in place and help communities build a culture of emotional support for older adults.
The Mary Free Bed project, titled MI-CHAP (Michigan Changes Healthy Aging Program) aims to evaluate an existing care navigation program to assess program success. The project also focuses on identifying areas for improvement for patients who are referred and discharged from Mary Free Bed following a stroke.
Patients enrolled in this program evaluation will work with care navigation specialists and researchers to rate their experience and to participate in a focus group on program enhancements. To learn more about this project, email research@maryfreebed.com.
Institutional Review Board
Mary Free Bed Rehabilitation operates its own Institutional Review Board (FWA: 00009374). For inquires regarding submission and approval, email irbadmin@maryfreebed.com.
Learning Health System
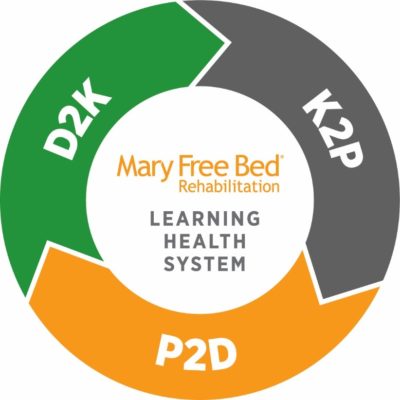 Mary Free Bed is committed to becoming a Learning Health System, an organization that engages and includes all workforce members to improve clinical and administrative processes with the goal of creating beneficial outcomes for patients. Our goal is to have a fluid system for reporting, evaluating, trialing, engaging and implementing from all corners of the Mary Free Bed Rehabilitation Network.
Mary Free Bed is committed to becoming a Learning Health System, an organization that engages and includes all workforce members to improve clinical and administrative processes with the goal of creating beneficial outcomes for patients. Our goal is to have a fluid system for reporting, evaluating, trialing, engaging and implementing from all corners of the Mary Free Bed Rehabilitation Network.
This iterative system will build on strengths already associated with Mary Free Bed: data warehouse, experienced and compassionate workforce, and engagement in the communities we serve.
To learn more, email research@maryfreebed.com.
Researchers
Research Education and Learning Health System Education modules are available on the Mary Free Bed Education Academy page here. These modules are intended to provide foundational knowledge related to conducting research and translating that research into practice.


